Potential enhanced fixation of hinged knee prostheses by additional zonal fixation and support in the metaphyseal knee area
Dr. Morgan-Jones / Zonal fixation in revision total knee arthroplasty
https://pubmed.ncbi.nlm.nih.gov/25628273/
Dr. Gehrke / Preliminary clinical results of coated porous tibia cones in septic and aseptic revision knee arthroplasty
https://link.springer.com/article/10.1007/s00402-020-03434-2
The purpose of the FlexiCones is to provide support in cases of bone loss, with the objective of stabilization in the distal femur or proximal tibia.
The selected cone is designed to:
- Strengthen the metaphyseal / diaphyseal medullary canal of the distal femur / proximal tibia
- Adequately fill the tibial/femoral bone cavity
- Ensure that assembled components are securely seated, e.g. femoral cone to femoral knee component, while at the same time supporting osteoconduction at the external surface of the cone
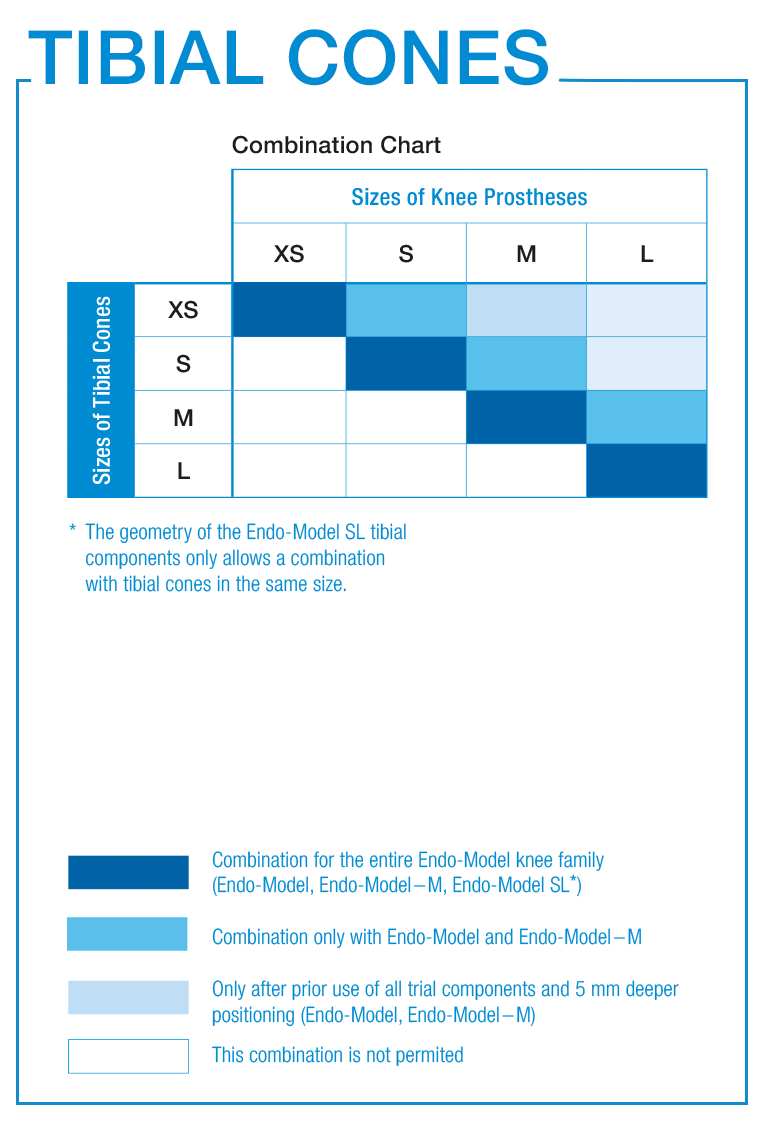
Both the TrabecuLink Femoral and Tibial Cones allow for a compliant combination with the entire Endo-Model knee family. In regulatory terms this is considered an on-label use of this combination.
On the other hand, combining our Endo-Model knee family with cones from competitors is considered an off-label use. In these combinations, the surgeon becomes the regulatory manufacturer of this combination.
Both the compensator and the patented bending axes allow the TL Cones to provide flexibility within the cone design. During the development phase, it was found that having built-in flexibility is an advantage. Compared to rigid cones, we found less stress when performing the final seating of the FlexiCones. The introduction of a rigid cone, on the other hand, resulted in bone fissures or even cracks because of an inhomogeneous load transfer.
After implantation, the FlexiCones follow the intramedullary nailing principle: Küntscher's concept of transverse elastic deadlock which can only be achieved by designing the cross-section of the implant itself to be elastic.
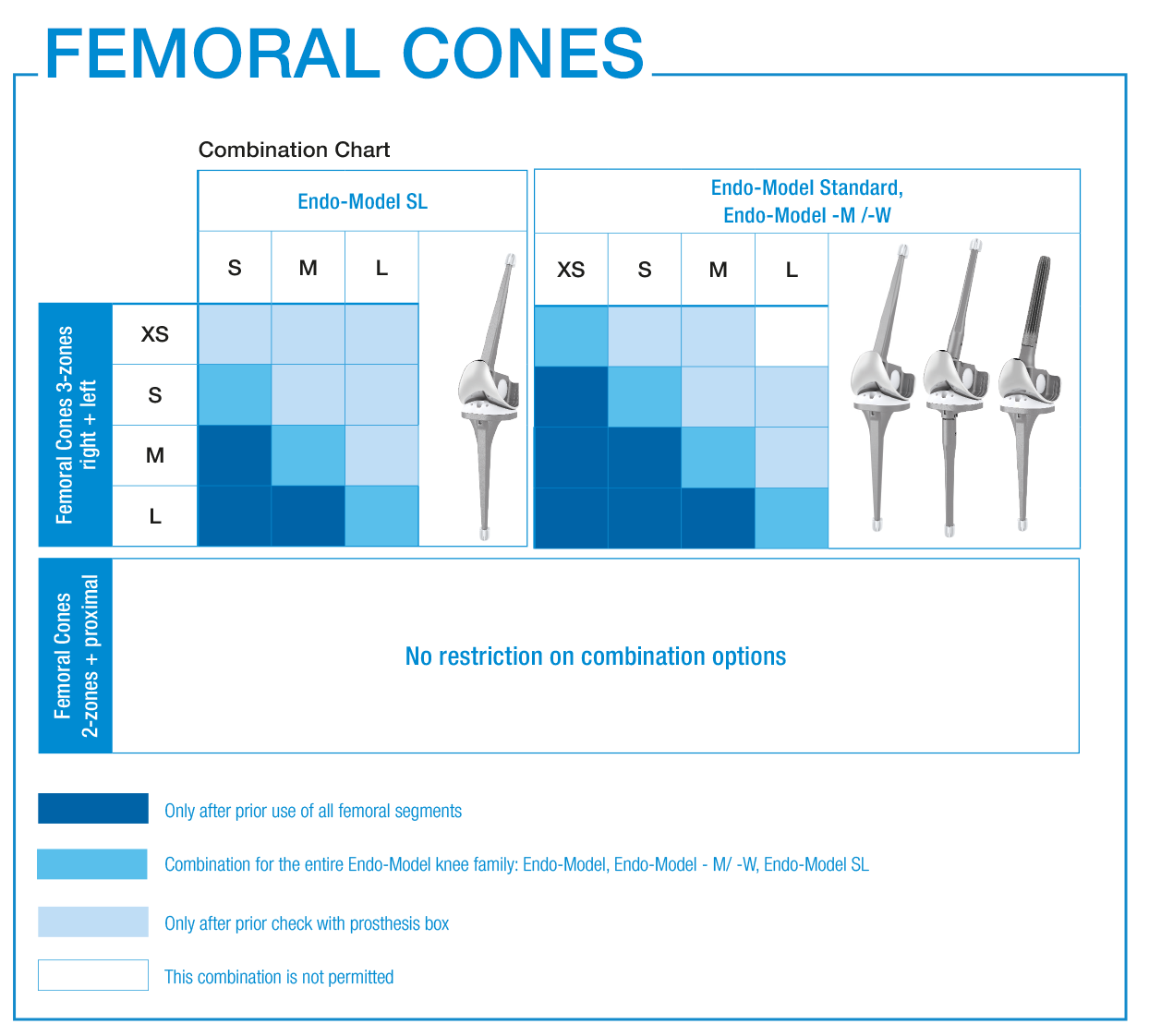
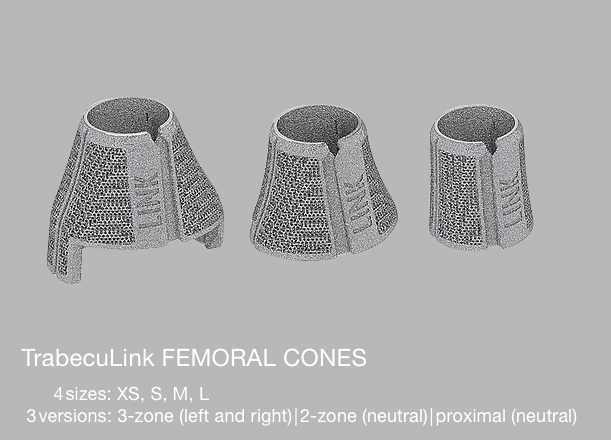
LINK offers 3-zones, 2-zones and proximal TrabecuLink Femoral Cones.
metaphyseal
- The 3-zone Femoral Cones cover the prosthesis box of all Endo-Model femoral components which are to provide additional rotational stability. In accordance with the Endo-Model femoral component, which is available in right/left versions, we also offer the Femoral Cones in right/ left versions.
- The 2-zone Femoral Cones are dimensionally identical, except for the absence of “legs”. Therefore, these Femoral Cones can be implanted side-neutral. Intraoperative positioning offers more freedom as there are no restrictions on the combination used with the Endo-Model femoral components.
diaphyseal
- The proximal Femoral Cones are used in the metaphyseal/diaphyseal area of the femur and are available as a side-neutral version.
LINK recommends always perfoming a test of all trial components, i.e. the trial femoral component, trial stem, trial segment, trial tibial component. After implantation of the Femoral Cone, we recommend double-checking that the FlexiCone is correctly seated and that the stem of the femoral component still fits.
Technically, cementless/ cemented stems with diameters of up to 16 mm (cylindrical) and 18 mm (conical) can be combined.